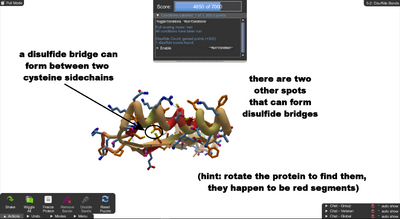
Disulfide Bonds. There's one at the start, find two more pairs of cysteines to make two more.
A condition in this puzzle awards a bonus for disulfide bonds, also known as disulfide bridges.
Loci's "Disulfide Bonds" video
Disulfide bonds are shown as yellow-and-green "candy canes" or spirals.
When the puzzle starts, there is already one disulfide bond. To solve the puzzle, you'll need to form two more disulfide bonds.
You'll need to rotate the protein by dragging on the background. It may also help to zoom in on the protein by holding shift and dragging on the background.
First, rotate and zoom to get a good view of the existing yellow-and-green spiral. It occurs between the tips of two cysteine sidechains. You may notice that the tips are yellow, indicating they are sulfur atoms. (It helps to hover over one of the sidechains, which brings out the yellow.)
"Disulfide" means "involving two sulfur atoms".
The goal is is to find more cysteine sidechains. You'll find there are two spots with pairs of sidechains near each other. In this puzzle, you'll be able to form an additional disulfide bridge easily just by dragging the tip of one of the cysteines so that it points toward the other cysteine in the pair.

Two cysteines near each other, but no disulfide bridge.
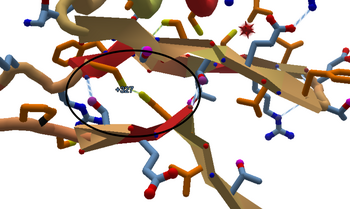
Two cysteines bonded in a disulfide bridge.
Technical stuff: disulfide bonds are much stronger than hydrogen bonds. Disulfide bonds occur between the sulfur atoms at the tips of two cysteines. The two cysteine segments must be aligned correctly for a bond to form. In a disulfide bond, the sidechains of the two cysteines are at nearly right angles to each other, and the sulfur atoms are about 2 Angstroms apart.
Because disulfide bonds are so strong, they can pose a problem for proteins found inside a cell. Cells need a way to recycle these intracellular proteins when they're not needed. The more disulfide bonds, the more difficult it would be to break down the protein.
For proteins which end up outside a cell, recycling is not an issue. Proteins in hair, fingernails, hooves, and fur contain lots of disulfide bonds.