In theory, every protein should have an inside, or core, and an outside, or surface. Most of the hydrophobic segments should be buried in the core. Most of the hydrophilic segments should be exposed on the surface.
In practice, real proteins may not follow these rules 100%.
The idea is that proteins at least start out inside cells, and cells are full of water molecules. The hydrophilic amino acids can stand up to the water molecules, but the hydrophobics need to be protected.
Blue sidechains want to be exposed outside and orange sidechains want to be buried inside.
When using the Core colors, this is the color scheme:
- Core (orange)
- Surface (blue)
- Boundary (green)
"Boundary" sidechains are marked as those that are close to being buried in the core.
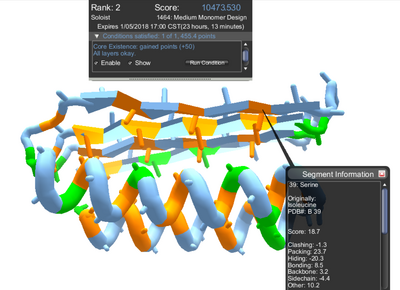
The "show" option of the core exists condition colors core segments in shades of orange. (Click for larger image.)
Most recent design puzzles include a core existence condition (formerly called a filter), which awards a bonus if the protein has a suitable core, or else subtracts a penalty. The exact requirements may vary, but a typical core existence condition might require that "at least 30% of the residues are buried in the core of your design".
The core exists condition was explained in more detail in a Foldit blog post by bkoep, which states:
since the protein is surrounded by water, the only way to hide the hydrophobic residues is to fold them into the center of the protein and shield them from water
The "show" of the core exists condition changes the coloring of the proten when checked. Core segments are shown in shades of orange. Surface segments are shown in blue. Segments on the boundary between core and surface are shown in green. Hydrophilic segments, such as serine are shown in a darker shade of orange when they're found in the core. (Unchecking the "show" option returns the protein to its previous coloring.)